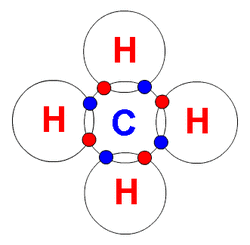
Ionic and covalent bonds are formed so an element complete the octet rule. The Octet Rule states that atoms bond to acquire eight electrons in it's outer shell. Ionic bonds are formed by the transfer of electrons between atoms. Covalent bonds are formed when two atoms share electrons. Ionic compounds are called salts. Ionic compounds are also called formula ratio. Cations and anions are held together by opposite charges through electrostatic attractions. Electrons are transferred to achieve noble gas configuration.
|
Properties of ionic compounds include:
|
Properties of covalent bonds
|

"Exploring Chemical Bonding." Welcome. N.p., n.d. Web. 28 May 2014.